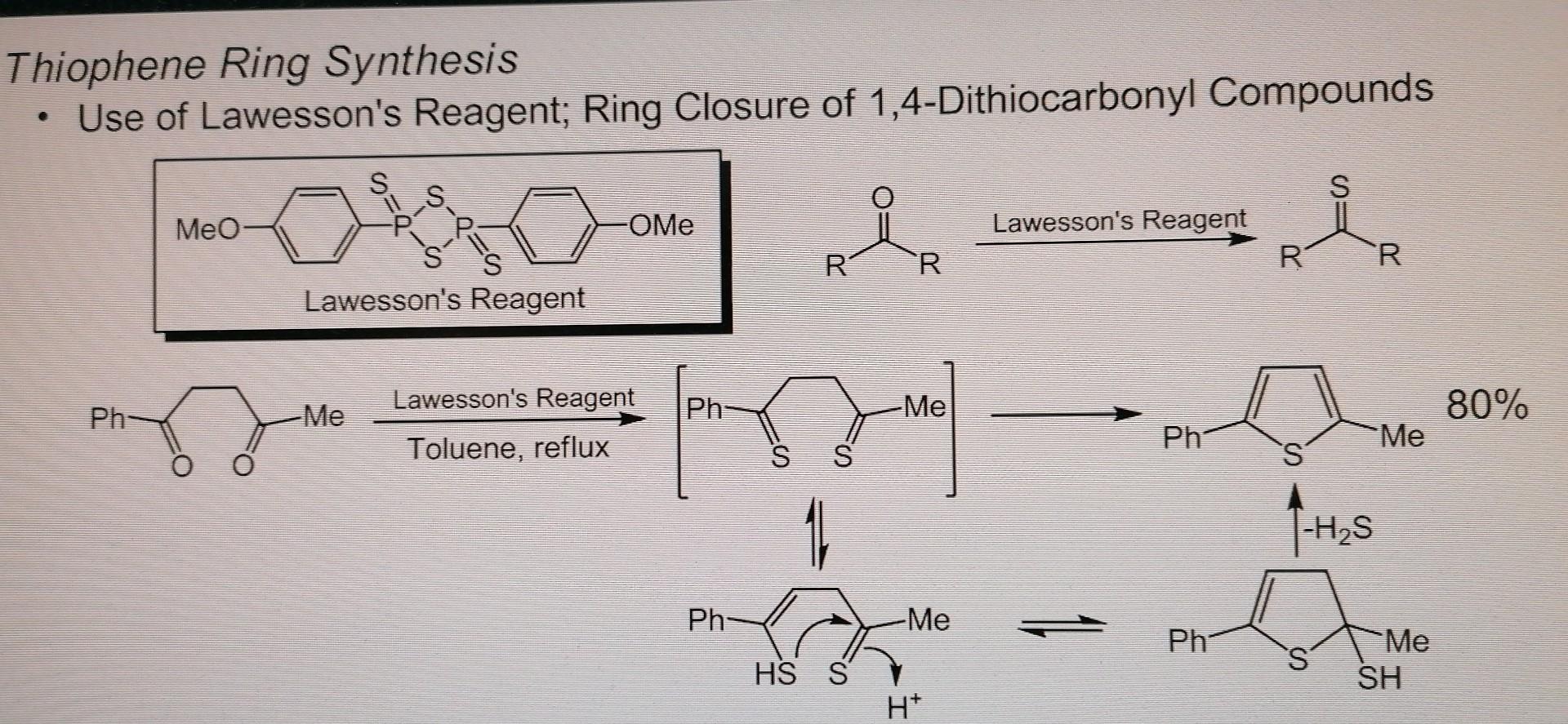
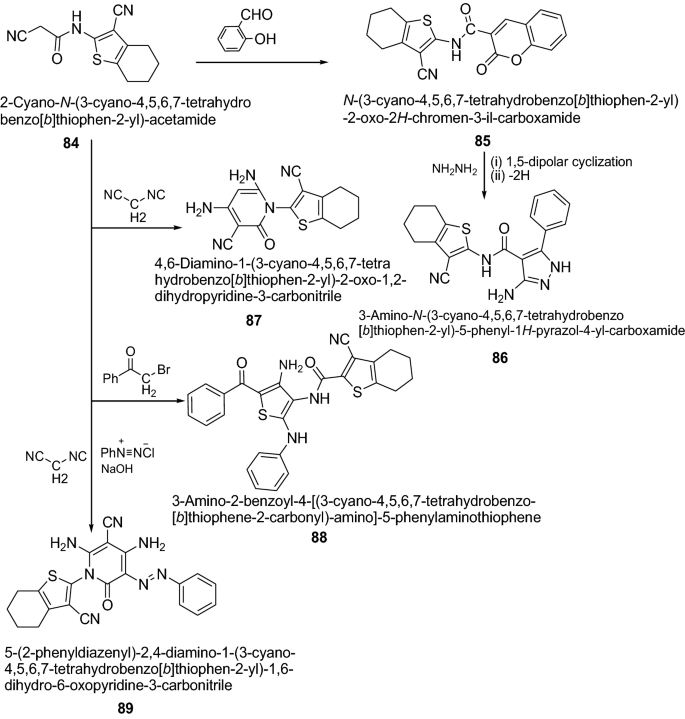
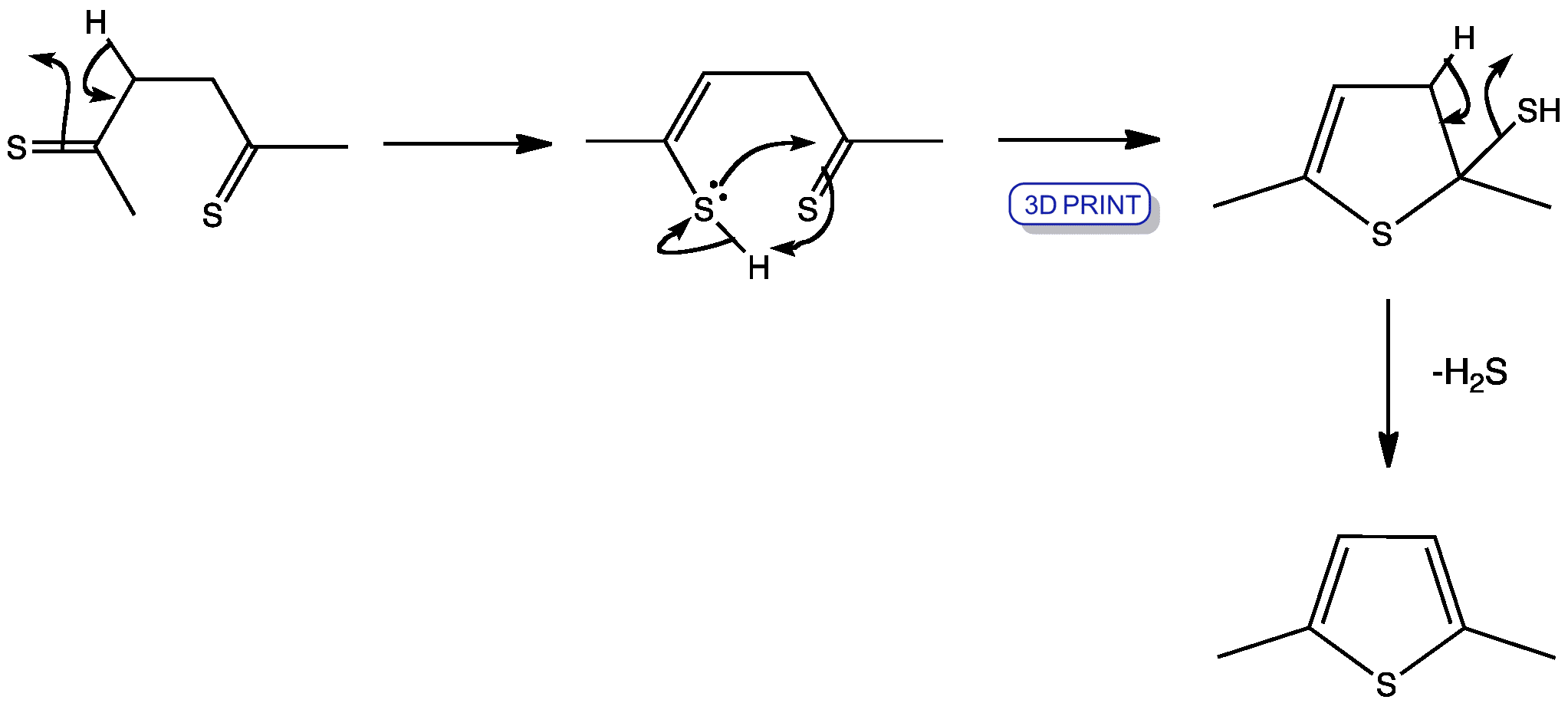
Ring-Expansion and Desulfurisation of Thiophenes with an Aluminium(I) Reagent | Inorganic Chemistry | ChemRxiv | Cambridge Open Engage
![Thiophene Ring-Opening Reactions V. Expedient Synthesis of 1,3,4- Thiadiazoline-Sulfanyl[1,3]thiazin-4-one Hybrids | Bentham Science Thiophene Ring-Opening Reactions V. Expedient Synthesis of 1,3,4- Thiadiazoline-Sulfanyl[1,3]thiazin-4-one Hybrids | Bentham Science](https://www.eurekaselect.com/images/graphical-abstract/loc/19/11/005.jpg)
Thiophene Ring-Opening Reactions V. Expedient Synthesis of 1,3,4- Thiadiazoline-Sulfanyl[1,3]thiazin-4-one Hybrids | Bentham Science

Functionalization of thiophene ring (left) and general structures of... | Download Scientific Diagram

Thiophene ring-opening reactions. Direct access to the synthesis of 1,3,4-thiadiazoline-(condenced) pyridone hybrids - ScienceDirect

The structure of terthiophene; three thiophene rings are arranged in an... | Download Scientific Diagram

Metallole, raney Nickel, Isatin, thioether, mw 3, thiophene, crc, pyrrole, furan, Heterocyclic compound | Anyrgb
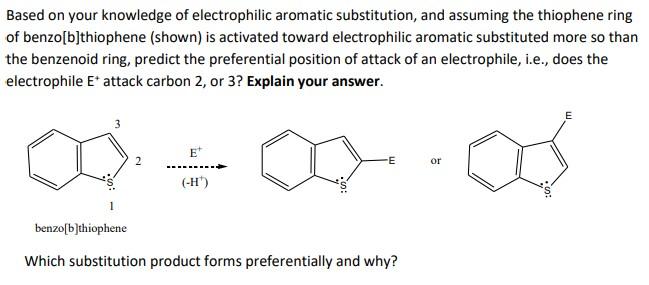
![SOLVED: Based on your knowledge of electrophilic aromatic substitution, and assuming the thiophene ring of benzo[b]thiophene (shown) is activated toward electrophilic aromatic substitution more so than the benzenoid ring, predict the preferential SOLVED: Based on your knowledge of electrophilic aromatic substitution, and assuming the thiophene ring of benzo[b]thiophene (shown) is activated toward electrophilic aromatic substitution more so than the benzenoid ring, predict the preferential](https://cdn.numerade.com/ask_images/c6f1484fa17f4780becf66aea189d7fd.jpg)
SOLVED: Based on your knowledge of electrophilic aromatic substitution, and assuming the thiophene ring of benzo[b]thiophene (shown) is activated toward electrophilic aromatic substitution more so than the benzenoid ring, predict the preferential

Aromaticity Heterocyclic compound Sulfur Thiophene Oxazole, aromatic ring, angle, white, text png | PNGWing

Thiophene ring-opening reactions. Direct access to the synthesis of 1,3,4-thiadiazoline-(condenced) pyridone hybrids - ScienceDirect
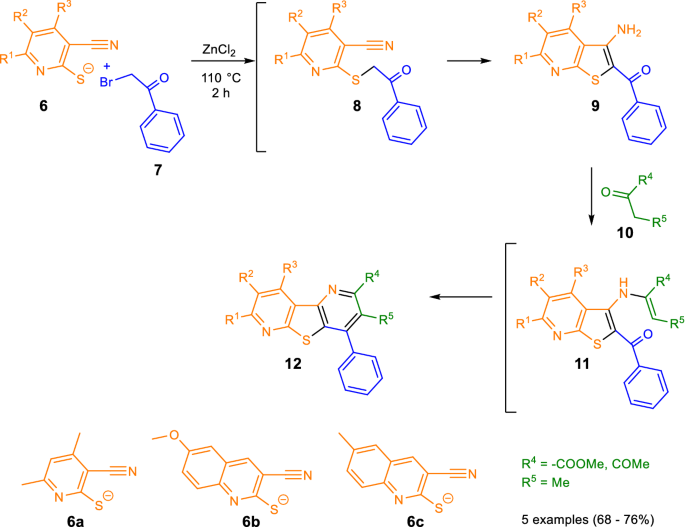
![Thiophene ring-opening reactions VI. Attempted cyclization towards [fused]-tricyclic system involving a thiolate anion and suitably located electrophilic carbon Thiophene ring-opening reactions VI. Attempted cyclization towards [fused]-tricyclic system involving a thiolate anion and suitably located electrophilic carbon](https://www.degruyter.com/document/doi/10.1515/znc-2022-0080/asset/graphic/j_znc-2022-0080_scheme_003.jpg)
Thiophene ring-opening reactions VI. Attempted cyclization towards [fused]-tricyclic system involving a thiolate anion and suitably located electrophilic carbon