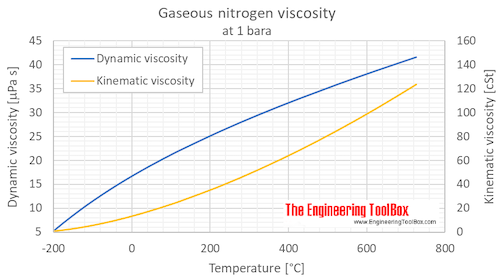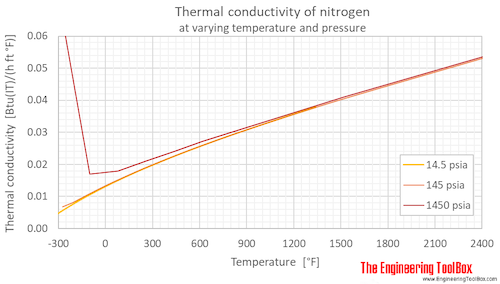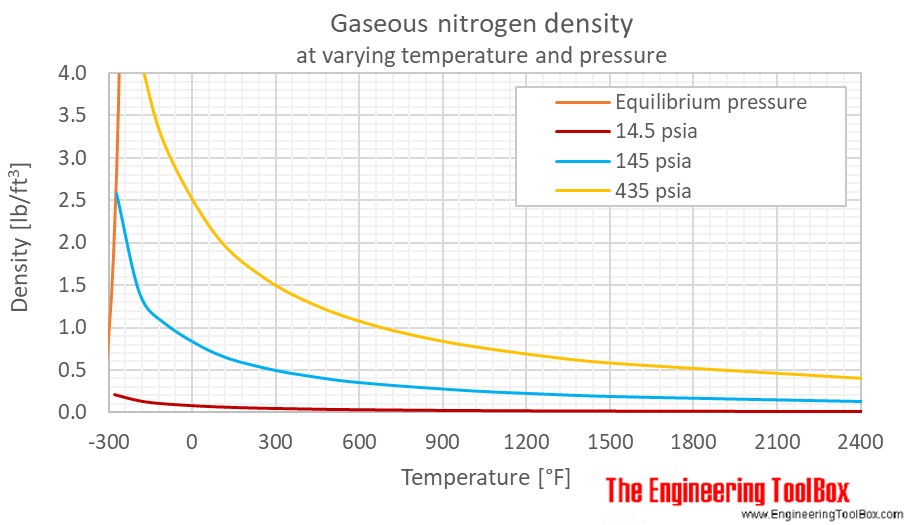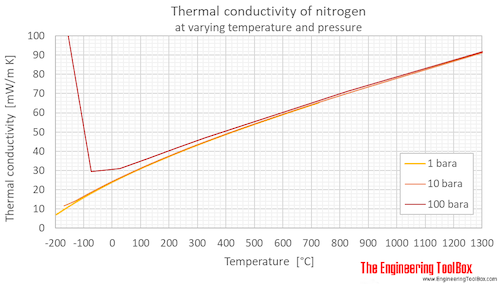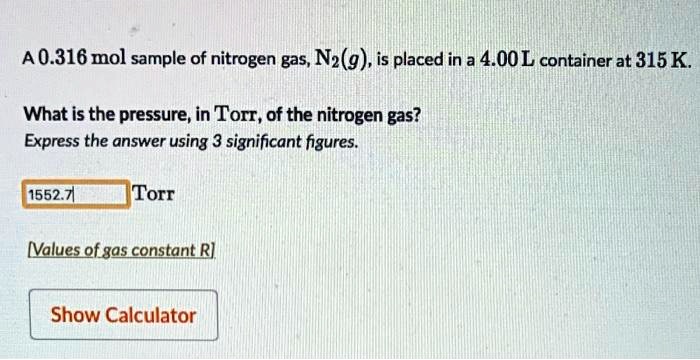
SOLVED: A 0.316 mol sample of nitrogen gas, N2(g), is placed in a 4.00 L container at 315 K. What is the pressure, in Torr, of the nitrogen gas? Express the answer

KALLER Gas Spring Calculators and Configurators | KALLER | World-leading brand for gas springs & hydraulic systems
Packaged Nitrogen Gas / Containers / Items / [SCIM] Satisfactory - Calculator | Gaming Tool/Wiki/Database to empower the players.
Nitrogen Gas / Gas / Items / [SCIM] Satisfactory - Calculator | Gaming Tool/Wiki/Database to empower the players.